We recently purchased a clock and the instructions stated that we must use a Zinc Carbon battery and not alkaline or any rechargeable battery, as the latter could damage the clock mechanism.
I have never heard of this phenomenon before - so is it real or just hype?
Is it something Choice can research?
pH
My guess (only that) is that some types of batteries are higher voltage than zinc-carbon, this article explains it:
For many years, the typical AA battery voltage was 1.5 volts with many devices still accepting only 1.5V AA batteries.
Lithium batteries that commonly feature 3.0+ volts, should NOT be used instead of 1.5V AA batteries unless explicitly allowed by the device manufacturer.
So if you check the voltage carefully other than zinc-carbon should be OK if they comply.
Standard single zinc carbon cells are nominally 1.5 volts. The same voltage as a similar single alkaline cell. Both will do the job.
Note:
https://www.panasonic-batteries.com/en/news/alkaline-batteries-versus-zinc-batteries
It’s important for low power items such as a clock to use a battery that has leak protection. Cheaper batteries of less well known brands of either chemistry will often start to leak as they approach flat.
Some further content. Choice has reviewed rechargeable batteries, and some time past non rechargeable batteries. You will need to log in as a member and search the Choice consumers web site.
P.S.
We use rechargeable NiMH in most devices that take standard AA and AAA batteries. I can’t recollect advice not to use alkaline in place of zinc carbon batteries with any product.
I can only presume that in a very low drain device like a clock, the battery would last years before running out of charge. Over time the battery could start to leak out its electrolyte, and that could cause damage.
But both zinc carbon and alkaline batteries can leak, so why one and not the other, who knows.
From the review:
There are a few online sites offering rechargeable Li-Ion AA batteries. Most of these batteries have a nominal voltage of 3.7V, which will fry your electronic devices if they take normal AA batteries, so avoid them unless you know exactly what devices you have that can cope with a nominal voltage greater than 1.5V.
I’ve had a couple of items which said “Don’t use Rechargeables” - which I’ve ignored, and they’re still working fine.
Maybe if it was an expensive item, I might be hesitant - but a clock.
I would like to offer another suggestion here, and that is that confusion has entered into the instructions in the process of translating from the original language into English.
I often come across crazy nonsensical statements in Asian instructions which have been transposed into English.
The only type of battery to avoid, as has already been mentioned, is lithium ion rechargeable batteries which have a terminal voltage as high as 4.2v when freshly charged. That is nearly 3 times the voltage of a standard zinc/carbon or alkaline cell and may indeed damage the clock mechanism.
BB.
“Which batteries should you use in low drain appliances such as a TV remote control or a clock? And which ones are ideal for your dect phone? Do you have to choose the zinc batteries or are the alkaline cells better? But what is the main difference…”
Zinc carbon batteries are fine for TV remotes which are a low drain intermittent use device.
But make it a habit to replace the batteries annually, even if they appear to be still working OK, as they may leak in the meantime and destroy your remote/clock/device.
Not quite the only ones. Rechargeable NiMH and NiCd batteries have a terminal voltage of 1.2V, which is not enough to power some devices specifying 1.5V cells when either 1.5, 3, 4.5, 6 etc voltage is required.
Lithium, alkaline or Zinc Carbon batteries rated at 1.5V should all be perfectly suited, the former 2 probably moreso for most devices due to their greater energy density and longer calendar life.
Actually they are the only type to avoid “in this case” because using a battery with a higher terminal voltage than approx 1.5v is the issue which may damage the clock, into which these batteries are to be installed.
Using the rechargeable type you mention, which have a lower voltage, is unlikely to result in damage to the clock mechanism, although quite possibly in some cases, the lower voltage of 1.2v may not be enough to work some devices properly.
BB
I think your previous suggestion is closer to the mark.
Just stupid user instructions.
A battery at 1.2 volts vs 1.5 volts is not going to damage anything.
And neither should a battery at 4 volts if the device has a power regulator, and those are just built in on a chip even on something cheap like a clock.
I reckon it is about leakage if batteries are left in low drain devices over years.
From what I’ve seen of the innards of those “quartz” clocks which use a 1.5v battery, there is no real electronics inside - just a small electro-magnet which tensions a spring every now and then. The spring tension runs the clock mechanism. When the tension runs low, the magnet operates for a fraction of a second to re-tension the spring and keep the clock running. There’s no “quartz crystal” anywhere to be seen! Some “quartz” watches use the same system.
With a duty cycle of about 100 milliSeconds every few minutes, an over voltage battery possibly won’t harm the device at all, but I won’t advise anyone else to take the risk.
While we’re on this topic it may be prudent to highlight the cheap “unknown brand” batteries that are often supplied with consumer electronic devices. Although these batteries seem like a great “freebie” they weigh in at about half the weight of brand name batteries, and they don’t last very long before they start leaking and wreck your device. I make it a habit to toss these batteries straight into the battery recycle box rather than risk installing them into my brand new device only to find they have leaked 4 months later and wrecked the device.
BB
Quartz? It has been timer ICs for decades now. If there is a quartz crystal it will be tiny and inside a very small IC. It may just be a timer chip like the venerable old 555s.
Of mice and men and clocks and chips and crystals.
Without pulling down a particular battery analogue dial clock drive it’s an open question. Some may offer a lesser standard of product, especially the 10 for $32.98 offers on eBay.
Reputable suppliers - well Jaycar, Bunnings, Clockworks, CWS and others all sell battery powered kits for clocks with hands. 1.5V AA or larger battery powered and Crystal locked electronics for accurate time keeping.
How long will a typical AA battery last and can low or high voltage damage the electronics ir mechanism? There is a general lack of specification.
Assume a quality product with a low voltage 2 phase stepper motor drive, H1344 or related IC and 32.768kHz precision cut/trimmed crystal. 1.2-1.8V appears to be a commonly stated operating voltage range for the IC.
1.2 volts is the nominal voltage of a NiMH rechargeable battery IE output voltage at 50% discharge. Marginal but perhaps it will be adequate in a typical quartz clock drive. Which may answer one part of the question.
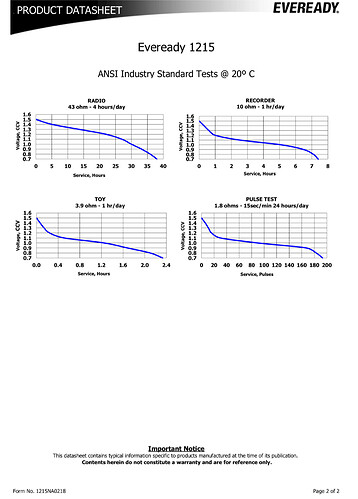
Focus on 1.5V from a typical AA Carbon zinc battery and the discharge curves typical of the breed (carbon zinc). Note that to get the most out of the Eveready black battery it needs to discharge to less than 1.0V IE the 1.5V commonly referred to is a nominal value when fresh. (I’ve typically measured 1.55-1.65V open circuit for unused new batteries.)